Breast cancer is the most prevalent cancer in women in the United States, accounting for 15.3% of all new cancer cases.1 In 2020 alone, 276,480 women are estimated to be diagnosed with breast cancer and an estimated 42,170 women will die from this disease. Overall, the 5-year relative survival rate for women with breast cancer is 90%; however, the 5-year survival rate drops to 28.1% for patients with metastatic disease.1
Triple-negative breast cancer (TNBC)—breast cancer that is negative for estrogen receptor, progesterone receptor, and human epidermal growth factor (HER)2—accounts for approximately 10% to 15% of all breast cancers.2,3 TNBC is more aggressive and has a poorer prognosis than other types of invasive breast cancer.3 In fact, women with TNBC whose disease is metastatic at diagnosis have a 5-year relative survival rate of only 11%.3
TNBC affects a substantial number of patients at an earlier age than other types of breast cancer.4 An analysis of data from a national cancer database from 2010 and 2011 showed that of 38,813 patients with TNBC, 21,935 (56%) were aged ≤60 years and 11,512 (29%) were aged ≤50 years.4
TNBC does not respond to some of the effective treatments available for breast cancer, including hormone therapy or HER2-targeted therapies (eg, trastuzumab) and endocrine therapies (tamoxifen or aromatase inhibitors).2,5 Sequential chemotherapy represents the cornerstone of treatment for metastatic TNBC.6 Other treatments for metastatic TNBC include poly (ADP-ribose) polymerase (PARP) inhibitors and immunotherapy.2
There remains a marked need for effective treatment options for patients with metastatic TNBC, particularly given the aggressive nature of the disease and its current survival rate.6 Recently, a novel targeted therapy was approved by the US Food and Drug Administration (FDA) for the treatment of patients with metastatic TNBC.5
FDA Approves Trodelvy for Metastatic Triple-Negative Breast Cancer
On April 22, 2020, the FDA accelerated the approval of sacituzumab govitecan-hziy (Trodelvy; Immunomedics) for the treatment of adults with metastatic TNBC who received at least 2 previous therapies for metastatic disease.5 Sacituzumab govitecan, a Trop-2–directed antibody and topoisomerase inhibitor conjugate, was granted priority review for this indication. The FDA also granted sacituzumab govitecan a breakthrough therapy designation.5
“Metastatic triple-negative breast cancer is an aggressive form of breast cancer with limited treatment options. Chemotherapy has been the mainstay of treatment for triple-negative breast cancer. The approval of Trodelvy today represents a new targeted therapy for patients living with this aggressive malignancy….Today’s approval provides patients who’ve already tried 2 prior therapies with a new option,”5 said Richard Pazdur, MD, Director of the FDA’s Oncology Center of Excellence.
Mechanism of Action
Sacituzumab govitecan is composed of 3 components, including sacituzumab, a humanized monoclonal antibody, which binds to Trop-2; the drug SN-38, a topoisomerase I inhibitor that is toxic to cancer cells; and CL2A, a hydrolysable linker that links sacituzumab to SN-38.7
Trop-2 is a transmembrane calcium signal transducer that plays a role in regulating cell-to-cell adhesion. The expression of Trop-2 is associated with cancer growth, division, and spread in breast cancer, including TNBC. Similarly, the overexpression of Trop-2 can lead to the growth of cancer cells in epithelial carcinomas.8
Sacituzumab govitecan binds to Trop-2–expressing malignant cells and is internalized with the subsequent release of SN-38 via hydrolysis of the linker.7 SN-38 then stabilizes topoisomerase I-DNA covalent complexes, thereby preventing the religation of topoisomerase I–induced single-strand breaks and triggering apoptosis and cell death.7,8 In xenograft models of TNBC, sacituzumab govitecan reduced tumor growth.7
Dosing and Administration
Sacituzumab govitecan is available for injection as a 180-mg lyophilized powder in single-dose vials for reconstitution and is administered by intravenous infusion. Sacituzumab govitecan should not be administered as an intravenous push or bolus.7
Sacituzumab govitecan should not be substituted for or used with other drugs containing irinotecan or its active metabolite SN-38.7
The recommended dose of sacituzumab govitecan is 10 mg/kg once weekly on days 1 and 8 of continuous 21-day treatment cycles until disease progression or unacceptable toxicity. Patients should be premedicated for the prevention of infusion reactions and chemotherapy-induced nausea and vomiting.
Patients should also be monitored during the infusion and for at least 30 minutes after infusion completion. If adverse reactions occur, treatment interruption and/or dose reduction may be required.7
Pivotal Clinical Trial: IMMU-132-01
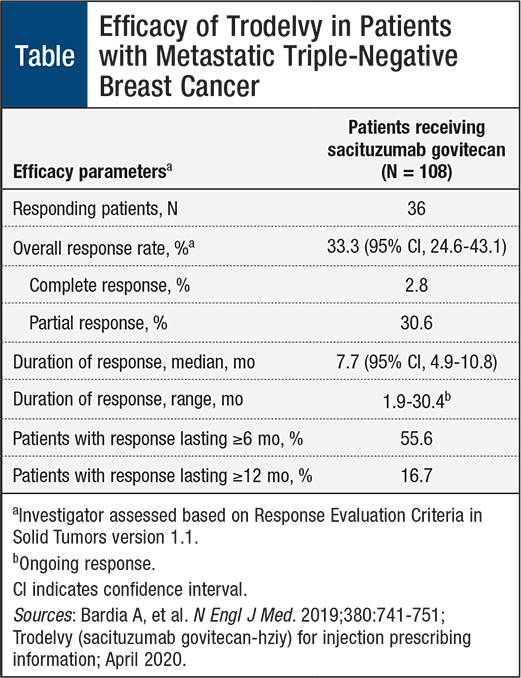
The approval of sacituzumab govitecan was based on efficacy results from the IMMU-132-01 trial, a single-arm, multicenter, phase 1/2 clinical trial that included 108 patients (median age, 55 years; range, 31-80 years) with metastatic TNBC who received at least 2 previous treatments for metastatic disease.6,7 Overall, 98% of patients previously received therapy with taxanes and 86% received treatment with anthracyclines either in the neoadjuvant or metastatic setting.7
Patients received sacituzumab govitecan 10 mg/kg intravenously on days 1 and 8 of a 21-day treatment cycle and continued treatment until disease progression or treatment intolerance. The key efficacy end points included overall response rate and duration of response.7
As shown in the Table, patients who received sacituz-umab govitecan achieved a 33.3% overall response rate and a median duration of response of 7.7 months. Moreover, 55.6% of the patients maintained a response lasting ≥6 months, and 16.7% maintained a response for ≥12 months.7
Adverse Events
The most common (≥25%) adverse reactions in patients with metastatic TNBC who received sacituzumab govitecan were nausea (69%), neutropenia (64%), diarrhea (63%), fatigue (57%), anemia (52%), vomiting (49%), alopecia (38%), constipation (34%), rash (31%), decreased appetite (30%), and abdominal pain (26%).7
Serious adverse events were reported in 31% of patients who received sacituzumab govitecan. Permanent treatment discontinuation resulting from adverse reactions occurred in 2% of patients.7
Drug Interactions
Sacituzumab govitecan should not be administered concomitantly with uridine diphosphate-glucuronosyl transferase 1A1 (UGT1A1) inhibitors or UGT1A1 inducers. The concomitant administration of sacituzumab govitecan with UGT1A1 inhibitors may increase the risk for adverse reactions that results from a potential increase in systemic exposure to SN-38, and the concomitant administration of sacituzumab govitecan with UGT1A1 inducers may substantially reduce exposure to SN-38.7
Contraindication
Sacituzumab govitecan is contraindicated in patients with a severe and life-threatening hypersensitivity to this drug.7
Use in Specific Populations
No data are available on the presence of sacituzumab govitecan or SN-38 in human milk or its effects on the breastfed child or on milk production. Lactating women should not breastfeed during treatment and for 1 month after the last dose of sacituzumab govitecan.7
No overall differences in safety and efficacy were observed between patients aged ≥65 years and younger patients.7
For patients with mild hepatic impairment, no adjustment to the starting dose is required when sacituzumab govitecan is administered. Data are not available on the safety of sacituzumab govitecan in patients with moderate or severe hepatic impairment.7
Warnings and Precautions
The prescribing information for sacituzumab govitecan contains a boxed warning about the risk for severe neutropenia and recommends monitoring the patient’s blood-cell counts periodically during treatment. Treatment should be withheld if the patient has an absolute neutrophil count of <1500/mm3 or a neutropenic fever. Treatment with a granulocyte-colony stimulating factor should be considered for secondary prophylaxis; anti-infective treatment should be initiated promptly if febrile neutropenia occurs.7
The boxed warning also cautions about the risk for severe diarrhea. Patients with diarrhea should be monitored and given fluid and electrolytes as needed, and atropine (if not contraindicated) should be administered for early diarrhea of any severity. If the patient has severe diarrhea, sacituzumab govitecan should be withheld until the diarrhea resolves to grade ≤1, and subsequent doses should be reduced. At the onset of late diarrhea, patients should be evaluated for signs of infection, and if negative, loperamide should be promptly initiated. In addition, patients should be provided with medications and instructions for the prevention and treatment of nausea and vomiting and premedicated with antiemetic drugs as a preventive measure.7
Hypersensitivity reactions, including severe anaphylactic reactions, have been observed with sacituzumab govitecan treatment. Patients should be monitored for infusion-related reactions. If severe or life-threatening reactions occur, sacituzumab govitecan should be discontinued permanently.7
Patients with reduced UGT1A1 activity (those who are homozygous for the UGT1A1*28 allele) have an increased risk for neutropenia after the initiation of sacituzumab govitecan treatment.7
Sacituzumab govitecan can cause fetal harm. Pregnant women and women of reproductive potential should use effective contraception during treatment and for 6 months after the last dose of sacituzumab govitecan.7
Conclusion
The recent approval of sacituzumab govitecan introduces a novel breakthrough targeted therapy for patients with metastatic TNBC—an aggressive form of breast cancer—that may improve outcomes for patients whose cancer did not respond to previous therapy and who have limited treatment options. In the IMMU-132-01 trial, sacituzumab govitecan, a Trop-2–directed antibody and topoisomerase inhibitor drug conjugate, demonstrated an overall response rate of 33.3% with a median duration of response of 7.7 months.
References
- SEER. Cancer stat facts: female breast cancer. https://seer.cancer.gov/statfacts/html/breast.html. Accessed June 1, 2020.
- National Cancer Institute. Breast cancer treatment (adult) (PDQ)—health professional version. Updated May 7, 2020. www.cancer.gov/types/breast/hp/breast-treatment-pdq#_1452. Accessed June 1, 2020.
- American Cancer Society. Triple-negative breast cancer. www.cancer.org/cancer/breast-cancer/understanding-a-breast-cancer-diagnosis/types-of-breast-cancer/triple-negative.html. Accessed June 1, 2020.
- Plasilova ML, Hayse B, Killelea BK, et al. Features of triple-negative breast cancer: analysis of 38,813 cases from the National Cancer Database. Medicine (Baltimore). 2016;95:e4614. doi: 10.1097/MD.0000000000004614.
- US Food and Drug Administration. FDA approves new therapy for triple negative breast cancer that has spread, not responded to other treatments. April 22, 2020. www.fda.gov/news-events/press-announcements/fda-approves-new-therapy-triple-negative-breast-cancer-has-spread-not-responded-other-treatments. Accessed June 2, 2020.
- Bardia A, Mayer IA, Vahdat LT, et al. Sacituzumab govitecan-hziy in refractory metastatic triple-negative breast cancer. N Engl J Med. 2019;380:741-751.
- Trodelvy (sacituzumab govitecan-hziy) for injection [prescribing information]. Morris Plains, NJ: Immunomedics, Inc; April 2020. www.trodelvy.com/pdf/PrescribingInformation.pdf. Accessed June 2, 2020.
- National Cancer Institute. NCI drug dictionary. Sacituzumab govitecan-hziy. www.cancer.gov/publications/dictionaries/cancer-drug/def/sacituzumab-govitecan. Accessed June 2, 2020.
Used with permission from American Health & Drug Benefits. Copyright © 2020 Engage Healthcare Communications.