Breast cancer is one of the most common cancer diagnoses in the United States. The National Cancer Institute estimates that 252,710 American women will be diagnosed with breast cancer and 40,610 will die from the disease in 2017.1 For the majority (62%) of women with breast cancer, the disease is confined to the breast, and the 5-year survival approaches 100%.1 However, for women with metastatic disease, the likelihood of survival at 5 years drops to 27%.1
Approximately 2 of 3 women with breast cancer have tumor cells that express estrogen and progesterone receptors.2 Patients with these hormone receptor (HR)-positive cancers are most often treated with hormone therapy. Depending on whether they are pre- or postmenopausal, women with advanced breast cancer may receive aromatase inhibitors, such as letrozole, anastrozole, and exemestane, or other estrogen-blocking drugs, such as toremifene, fulvestrant, and tamoxifen.2
Because postmenopausal women with estrogen receptor–positive breast cancer can live with their disease for several years, treatments that maximize efficacy and minimize toxicity are crucial. Recent clinical trials of combination therapies for women with HR-positive breast cancer have included antiestrogens paired with aromatase inhibitors, as well as aromatase inhibitors paired with targeted agents.3 In 2015, palbociclib (Ibrance; Pfizer), the first inhibitor of cyclin-dependent kinases (CDK) 4 and 6 (CDK4/CDK6), received accelerated approval for advanced breast cancer in postmenopausal women.4,5 Among patients who received palbociclib plus letrozole (Femara; Novartis), progression-free survival (PFS) was extended to 20.2 months compared with 10.2 months with letrozole alone.4,5
Ribociclib Approved for Advanced Breast Cancer
On March 13, 2017, the US Food and Drug Administration (FDA) approved ribociclib (Kisqali; Novartis), an oral CDK4/CDK6 inhibitor, in combination with letrozole for the treatment of postmenopausal women with HR-positive, HER2-negative advanced breast cancer.6,7 The FDA approval of ribociclib was based on results of a randomized study showing that ribociclib plus letrozole significantly improves PFS and overall response rate compared with letrozole plus placebo.6-8
In May 2017, the FDA approved the Kisqali Femara Co-Pack, a single package that includes ribociclib and letrozole.8
Mechanism of Action
Ribociclib is an inhibitor of the CDK4/CDK6 pathway. Upon binding to D-cyclins, these kinases are activated to affect signaling pathways that lead to cell-cycle progression and cell proliferation.7
Dosing and Administration
Ribociclib is administered in combination with letrozole. The recommended starting dose of ribociclib is 600 mg (three 200-mg oral tablets) for 21 days, followed by 7 days without treatment, resulting in a complete cycle of 28 days; letrozole 2.5 mg is taken once daily throughout this 28-day cycle. Ribociclib and letrozole should be administered at approximately the same time each day, preferably in the morning.7
Ribociclib can be taken with or without food; tablets should not be chewed, crushed, or split before swallowing.7
The MONALEESA-2 Pivotal Clinical Trial
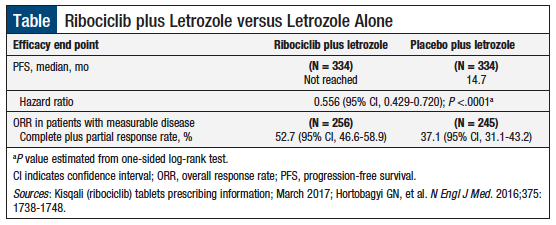
The efficacy and safety of ribociclib combined with letrozole was demonstrated in the randomized, double-blind, placebo-controlled clinical trial, MONALEESA-2.6,9 This multicenter study enrolled 668 postmenopausal women with HR-positive, HER2-negative advanced breast cancer.9 Patients were randomized in a 1:1 ratio to ribociclib (600 mg daily for 21 days in a 28-day cycle) plus letrozole 2.5 mg daily or to placebo plus letrozole 2.5 mg daily, and were stratified according to the presence of liver and/or lung metastases.9
The major efficacy outcome measure for the study was investigator-assessed PFS using Response Evaluation Criteria in Solid Tumors v1.1.9
All patients had an Eastern Cooperative Oncology Group performance status 0 or 1.9
At the interim analysis, PFS was not reached with ribociclib plus letrozole versus 14.7 months with placebo plus letrozole (Table).7,9 PFS and overall response rate results were consistent across patient subgroups.
The median exposure to ribociclib plus letrozole was 13 months, and the median follow-up from randomization was 15.3 months.9 After 12 months, the PFS rate was 72.8% (95% confidence interval [CI], 67.3-77.6) in the ribociclib arm versus 60.9% (95% CI, 55.1-66.2) in the placebo arm. After 18 months, the PFS rate was 63.0% and 42.2%, respectively.9
Dose reductions because of adverse events were required in 45% of patients in the ribociclib arm. In addition, 7% of patients in the ribociclib arm discontinued treatment because of adverse events versus 2% of patients in the letrozole plus placebo arm.9
The most common events leading to treatment discontinuation were increased alanine aminotransferase (4%) or aspartate aminotransferase (3%), and vomiting (2%). Common adverse events (frequency ≥20%) were neutropenia, nausea, fatigue, diarrhea, leukopenia, alopecia, vomiting, constipation, headache, and back pain.
The most common grade 3 or 4 adverse events were neutropenia, leukopenia, abnormal liver function tests, lymphopenia, and vomiting.9
Warnings and Precautions
Neutropenia has been observed in clinical trials of ribociclib.7
Ribociclib can prolong the QT interval in a concentration-dependent manner.7 Electrocardiograms should be repeated at approximately day 14 of the first cycle, and at the beginning of the second cycle, and as clinically indicated. Serum electrolytes should be assessed before the initiation of treatment, at the beginning of the first 6 cycles, and as clinically indicated. Any abnormality should be corrected before initiating ribociclib. Use of ribociclib with drugs known to prolong the QTc (corrected QT) interval and/or strong cytochrome P3A inhibitors should be avoided.7
Liver function tests should be performed before initiation of ribociclib, every 2 weeks for the first 2 cycles, at the beginning of each of the subsequent 4 cycles, and as clinically indicated.7
Women of reproductive potential should use effective contraception during therapy with ribociclib, and for ≥3 weeks after the last dose.7
Ribociclib has no contraindications.7
Use in Specific Populations
Based on animal studies, ribociclib can cause fetal harm when administered to a pregnant woman; no data are available in humans. Because of the potential risk, pregnant women should avoid using ribociclib.
Conclusion
Ribociclib is the second CDK4/CDK6 inhibitor to receive FDA approval. In the clinical trial that led to its approval by the FDA, adding ribociclib to letrozole significantly extended the PFS duration and improved the overall response rate in women with HR-positive, HER2-negative advanced breast cancer.
Disclaimer
The information in this article is current as of publication date and was derived from publicly available sources and the drug manufacturer’s website and prescribing information. The Publisher is not responsible for inaccurate information due to ongoing updates. Readers should refer to the drug manufacturer’s website for the most current information.
Copyright © 2017 American Health & Drug Benefits. Used with permission. All rights reserved.
References
- National Cancer Institute SEER program. Cancer stat facts: female breast cancer. http://seer.cancer.gov/statfacts/html/breast.html. Accessed June 15, 2017.
- American Cancer Society. Hormone therapy for breast cancer. Revised August 18, 2016. www.cancer.org/cancer/breastcancer/detailedguide/breast-cancer-treating-hormone-therapy. Accessed June 15, 2017.
- Inman S. Three experts examine the ER-positive treatment landscape. OncLive. February 26, 2015. www.onclive.com/conference-coverage/mbcc-2015/three-experts-examine-the-er-positive-treatment-landscape. Accessed June 23, 2017.
- US Food and Drug Administration. FDA approves Ibrance for postmenopausal women with advanced breast cancer. Press release. February 3, 2015. www.fda.gov/newsevents/newsroom/pressannouncements/ucm432871.htm. Accessed June 23, 2017.
- Ibrance (palbociclib) capsules [prescribing information]. New York, NY: Pfizer; March 2017.
- US Food and Drug Administration. Ribociclib (Kisqali). March 13, 2017. www.fda.gov/drugs/informationondrugs/approveddrugs/ucm546438.htm. Accessed June 15, 2017.
- Kisqali (ribociclib) tablets [prescribing information]. East Hanover, NJ: Novartis; March 2017.
- Toich L. Kisqali Femara co-packaging approved for breast cancer treatment. Pharmacy Times. May 9, 2017. www.pharmacytimes.com/news/kisqali-femara-copackaging-approved-for-breast-cancer-treatment. Accessed June 23, 2017.
- Hortobagyi GN, Stemmer SM, Burris HA, et al. Ribociclib as first-line therapy for HR-positive, advanced breast cancer. N Engl J Med. 2016;375:1738-1748.